About API
API Vietnam Company is one of Vietnam’s fastest-growing and pioneering firms, specializing in the import, distribution, and trading of Active Pharmaceutical Ingredients (APIs), Finished Dosage Form (FDFs), excipients, and fine chemical items (including food/feed ingredients and cosmetics).
Two Decades of Excellence: Built on a foundation of over 30 years of leadership insight in API and FDF sectors, and backed by nearly 20 years of sustainable growth, we are proud to be a leading strategic partner in Vietnam’s pharmaceutical industry.
Professional Team: Our experienced and robust team maintains strong client relationships with both international partners and domestic enterprises.
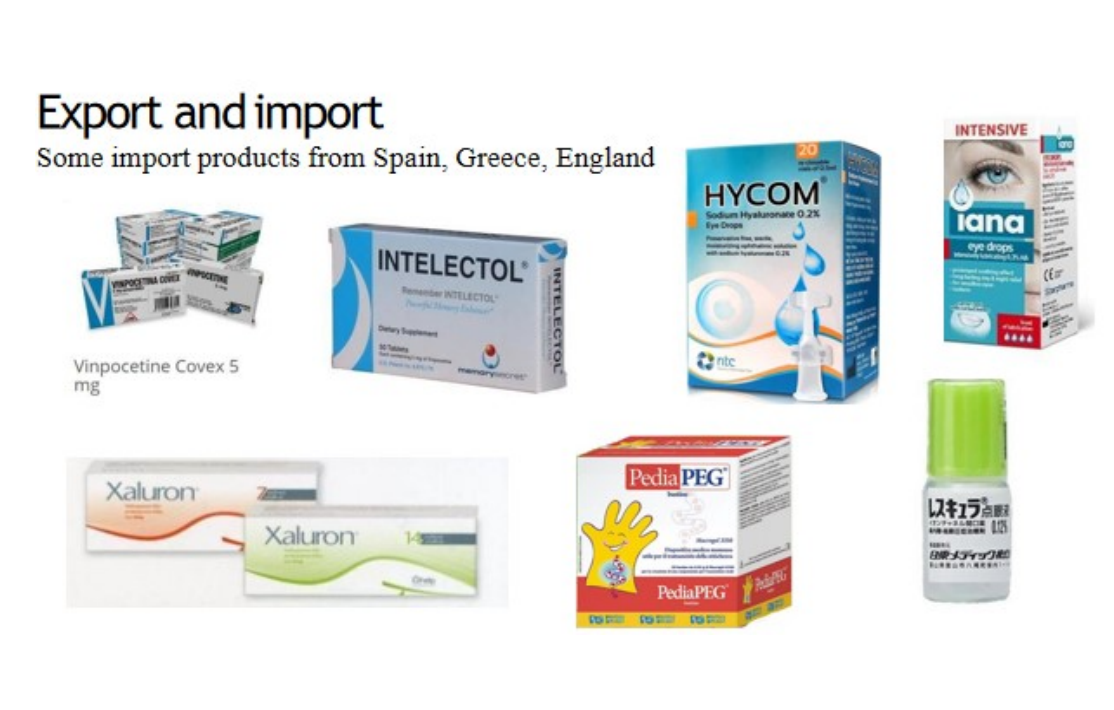
Premium Sourcing: We proudly represent reputable, high-quality foreign manufacturers, offering a diverse portfolio of over 1000 products. This includes specialized antibiotics (Cephalosporin, Penem, Penicillin) sourced from Europe, China, India, and more.
Guaranteed Compliance: All products adhere to the latest international standards and are backed by full GMP/DMF/CEP…. certifications from industry leaders such as Zhejiang, Hunan, ACS Dobfar, Moehs, Cambrex, Supriya, BDR, Welding…..